Principal investigator
Clinical research coordinator
Clinical operations manager
Regulatory affairs specialist
Sponsor study director
IRB coordinator

This process is used when a potential participant has been identified for a clinical study and must be evaluated for eligibility, consented, and formally enrolled in accordance with the study protocol, ICH-GCP guidelines, and regulatory requirements. It applies when screening assessments, eligibility criteria verification, informed consent documentation, and randomization or registration procedures must be completed accurately and within protocol-defined windows. It is common when investigators, coordinators, laboratory staff, sponsors, and the participant must coordinate across multiple screening activities. Ideal for pharmaceutical sponsors, CROs, academic medical centers, and clinical trial sites enrolling subjects in FDA-regulated or globally-regulated studies.
The enrollment process typically involves clinical research coordinators who manage the enrollment workflow and patient contact, principal investigators who verify eligibility and oversee the informed consent process, laboratory and diagnostic staff who perform screening assessments, sponsor study teams who may review eligibility queries and randomization, and the participant who provides informed consent and completes screening activities.
Compliant enrollment because every step — screening, eligibility verification, consent, and randomization — is documented in accordance with protocol and regulatory requirements. Fewer enrollment errors through structured eligibility checks that catch inclusion/exclusion violations before randomization. Faster screen-to-enrollment timelines by coordinating screening activities and consent in parallel where the protocol permits. Protected subject rights through documented informed consent processes that confirm voluntary, informed participation. Accurate enrollment data that supports trial integrity, site performance metrics, and sponsor reporting.
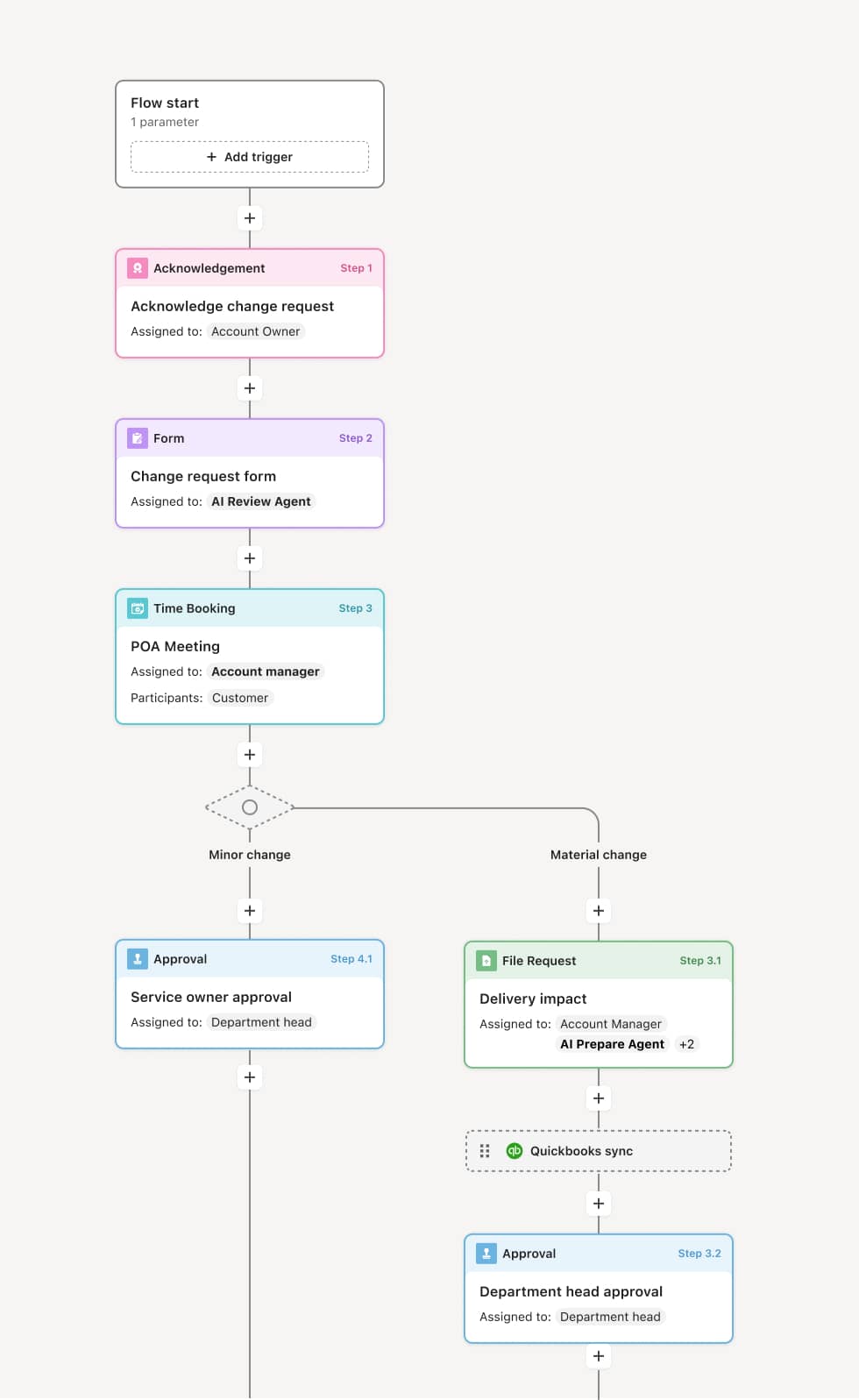
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo’s flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Pre-screening and identification
The process begins when a potential participant is identified through referral, patient database review, or self-referral. The coordinator performs a preliminary assessment against the study’s key inclusion and exclusion criteria to determine whether the individual is a reasonable candidate for formal screening. An AI Agent can assist by comparing available medical history against the protocol’s major eligibility criteria to flag potential disqualifiers early.
Informed consent
Before any study-specific screening procedures, the informed consent process is conducted. The investigator or qualified designee explains the study purpose, procedures, risks, benefits, alternatives, and the participant’s rights. The participant has the opportunity to ask questions and is given adequate time to decide. Written informed consent is obtained and documented. An AI Agent may verify that the most current IRB-approved consent document version is being used.
Screening assessments
The participant undergoes the protocol-specified screening assessments, which may include medical history review, physical examination, laboratory tests, imaging, and questionnaires. Results are collected within the protocol-defined screening window. If screening assessments are not completed within the required timeframe, the screening may need to be repeated or the participant may not proceed.
Eligibility verification
The investigator reviews all screening results against the protocol’s inclusion and exclusion criteria. Every criterion must be documented as met or not met. If the participant does not meet all criteria, they are classified as a screen failure and the reason is documented. If eligibility requires sponsor confirmation or central review, the verification is submitted for approval before proceeding. An AI Agent may cross-reference screening results against each criterion to flag potential eligibility issues.
Randomization or registration
Upon confirmed eligibility, the participant is randomized (for randomized studies) or registered into the study through the interactive response technology (IRT) system or sponsor portal. The assignment is documented and the participant formally becomes an active subject.
Enrollment confirmation and initiation of study activities
The participant is confirmed as enrolled, study materials are provided, and the first study visit or treatment is scheduled. The site, sponsor, and participant all have documented records of the enrollment. The complete enrollment record — consent, screening data, eligibility verification, and randomization — is preserved.
This process commonly relies on inputs such as the study protocol, informed consent documents, screening assessment results, inclusion/exclusion criteria checklists, and IRT randomization data. It may be triggered by a patient referral, a pre-screening match, or a self-referral inquiry. Connected systems often include EDC platforms like Medidata Rave or Oracle InForm, CTMS platforms for site and enrollment tracking, IRT systems for randomization, and EHR platforms for medical history data.
Key decision points include whether the potential participant passes pre-screening criteria to warrant formal screening, whether informed consent is properly obtained before any study-specific procedures, whether all inclusion criteria are met and no exclusion criteria are present, and whether sponsor confirmation or central eligibility review is required before randomization.
Consent obtained with an outdated document version, creating a protocol deviation and requiring re-consent. Screening assessments completed outside the protocol-defined window, invalidating results and requiring repeat testing. Eligibility criteria not fully documented, leaving gaps that create findings during monitoring or audit. Screen failures not properly documented, making it difficult to demonstrate that the enrollment process was followed. Randomization errors when the IRT system is accessed before eligibility is fully confirmed and documented.
Orchestrates the enrollment workflow from pre-screening through randomization across coordinators, investigators, laboratory staff, and sponsors in a single coordinated flow.
Ensures the correct consent document version is used by tracking IRB-approved versions within the workflow and flagging outdated documents.
AI Agents compare screening results against eligibility criteria to identify potential disqualifiers before the investigator’s final eligibility determination.
Tracks screening assessment timelines to ensure all procedures are completed within the protocol-defined screening window.
Connects to EDC, CTMS, and IRT systems like Medidata, Veeva Vault, and IRT platforms so enrollment data flows directly into study records and randomization.
Preserves the complete enrollment record including consent documentation, screening data, eligibility verification, and randomization for regulatory compliance, monitoring, and audit readiness.