Compliance officer
Risk manager
Regulatory affairs lead
Legal counsel
Operations manager
Quality assurance director

This process is used when business activities require compliance verification before execution. It applies when new products or services need regulatory clearance, when customer transactions exceed risk thresholds, when marketing materials require legal review, when vendor relationships need due diligence validation, or when operational changes impact regulated processes. Compliance approval is common in financial services, healthcare, pharmaceuticals, manufacturing, and any industry with significant regulatory oversight.
Participants typically include the business owner who submits the request for compliance review, subject matter experts who provide context on the activity or transaction, compliance analysts who evaluate against applicable requirements, and senior compliance officers who authorize approval or escalate concerns. Depending on the matter, legal counsel, risk management, or executive leadership may be involved in high-stakes decisions.
Regulatory confidence with documented verification that activities meet applicable requirements before proceeding. Risk mitigation by identifying compliance concerns early, before they become violations or exposures. Clear audit trails demonstrating that compliance review occurred and who authorized the activity. Faster business execution by streamlining compliance review rather than creating bottlenecks. Consistent policy application ensuring similar activities receive similar compliance treatment.
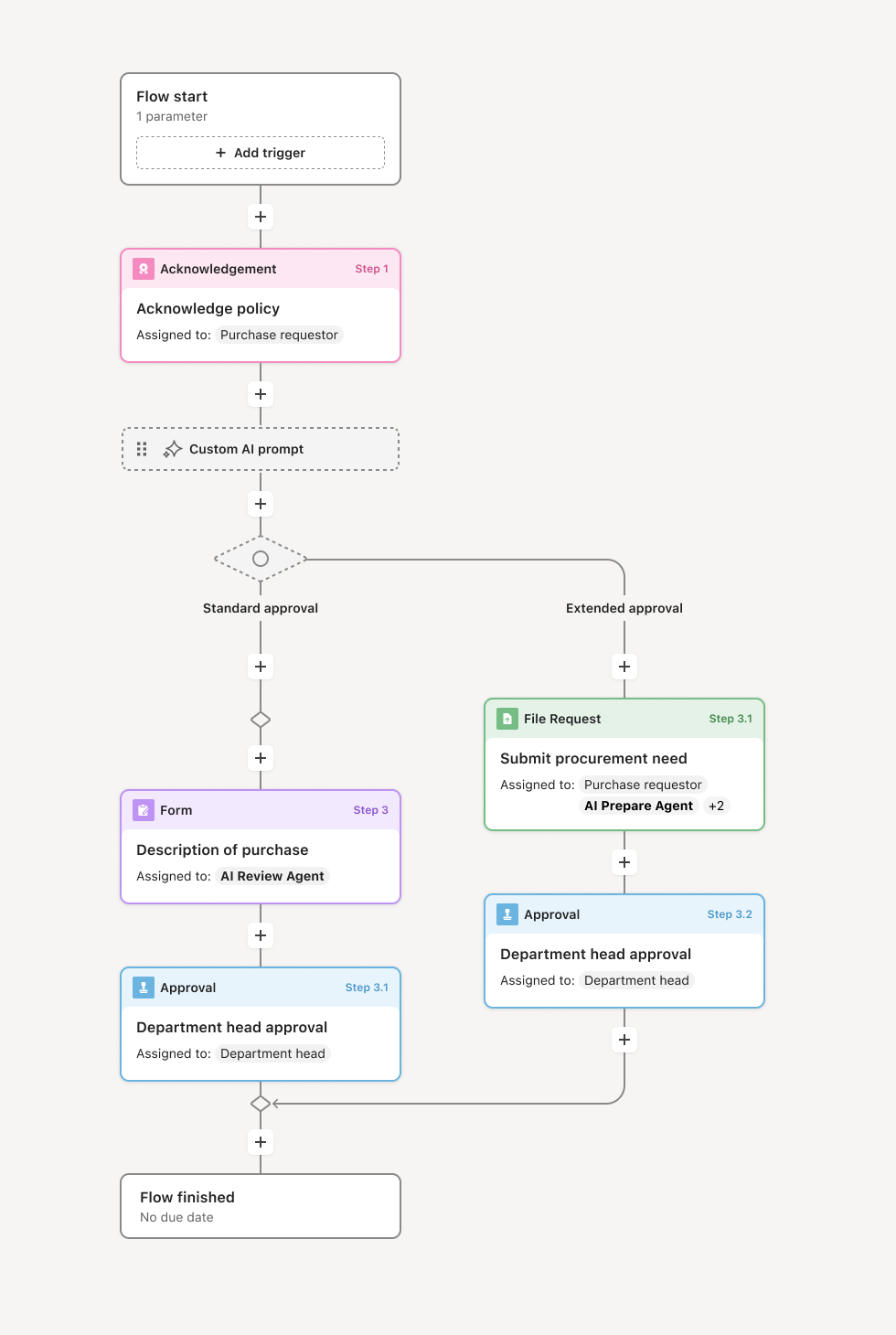
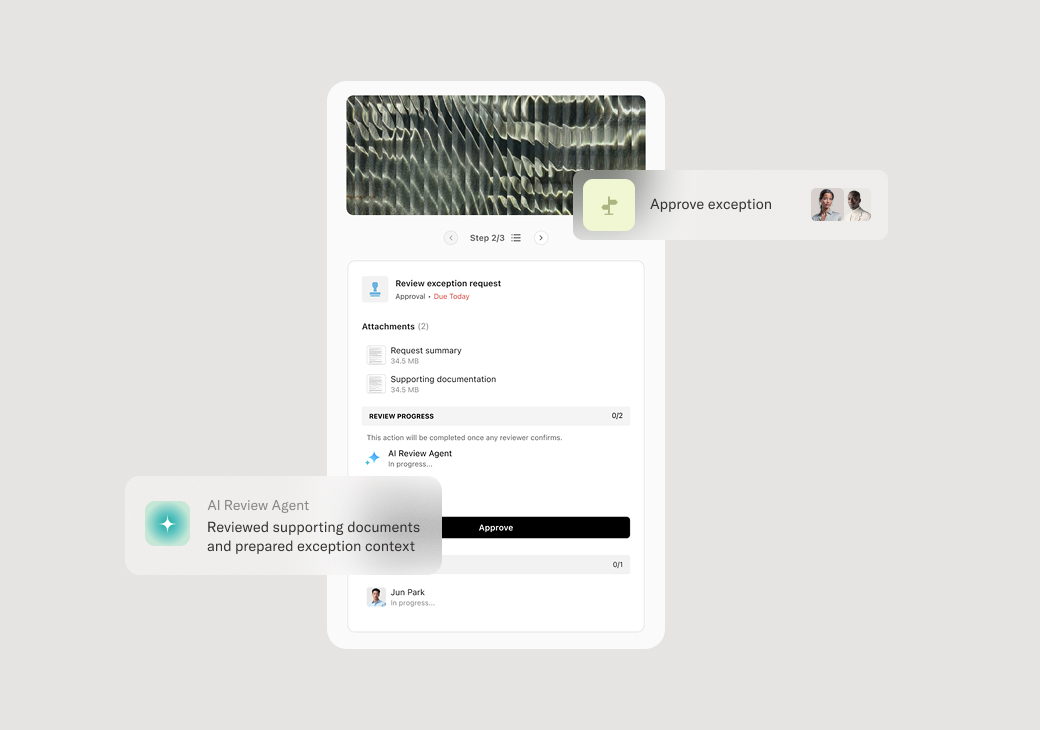
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo's flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Request submission and classification
The process begins when a business team submits an activity, transaction, or document for compliance review. The submission includes relevant details, supporting documentation, and context about the intended action. An AI agent may assist by classifying the request type, identifying applicable regulations, and routing to the appropriate compliance reviewer based on subject matter.
Requirement identification
The compliance team identifies which regulations, policies, and standards apply to the submitted request. This may include industry regulations, internal policies, contractual obligations, or jurisdictional requirements. The applicable requirements are documented to frame the review scope.
Compliance assessment
The compliance analyst evaluates the request against identified requirements, reviewing documentation, assessing risk factors, and identifying any gaps or concerns. If additional information is needed, the analyst requests clarification from the submitting team. AI agents may assist by checking submissions against policy databases or flagging common compliance issues.
Issue resolution and remediation
If compliance concerns are identified, the analyst documents the issues and works with the business team to develop remediation approaches. This may involve modifying the proposed activity, adding controls, obtaining additional approvals, or in some cases, declining to proceed. The resolution path is documented for audit purposes.
Approval and authorization
Once compliance requirements are satisfied, the request is approved and documented. For high-risk or complex matters, senior compliance officers or committees may provide final authorization. The approval record includes the scope of what was reviewed, applicable requirements, any conditions, and the authorizing parties.
Monitoring and follow-up
Depending on the activity type, ongoing monitoring requirements may be established. The workflow may trigger periodic reviews, reporting obligations, or renewal processes. Compliance records are maintained for audit and regulatory examination purposes.
This process commonly relies on inputs such as transaction details, product specifications, marketing materials, vendor documentation, or operational procedures. It may be triggered by events like deal submissions in a CRM, product launches in development systems, or manual requests through compliance portals. Supporting systems might include compliance management platforms, policy databases, risk assessment tools, and document management systems.
Key decision points include determining which requirements apply to the request, whether the submission satisfies those requirements, whether identified issues can be remediated, and whether residual risk is acceptable. If requirements cannot be met, the workflow branches to rejection or escalation paths. If conditional approval is appropriate, the conditions are documented and tracked.
Incomplete submissions that lack sufficient information for compliance assessment, causing delays. Unclear requirement mapping when applicable regulations are not properly identified, leading to gaps in review. Bottlenecks in review when compliance teams are overwhelmed and requests queue without timely attention. Undocumented exceptions when deviations from standard requirements are approved without clear rationale.
Structures compliance submissions so reviewers receive complete information and documentation upfront.
Routes requests to appropriate reviewers based on subject matter, risk level, or regulatory domain.
AI agents assist with classification and validation by identifying applicable requirements and flagging common issues.
Tracks review status and deadlines with automated reminders to prevent requests from stalling.
Captures complete approval records including requirements reviewed, issues addressed, and conditions imposed.
Integrates with compliance management systems to maintain centralized records and connect to ongoing monitoring workflows.