HR compliance manager
Occupational health coordinator
Medical review officer
Safety director
Hiring manager
Workplace health program manager

This process is used when the organization requires drug testing for pre-employment screening, random testing programs, post-incident testing, reasonable suspicion testing, or return-to-duty protocols. It applies when the testing must comply with federal regulations (such as DOT or SAMHSA guidelines), state laws, or organizational policies, and when chain-of-custody, specimen handling, and result confidentiality must be maintained throughout. It is common when HR, occupational health, collection sites, laboratories, and medical review officers must coordinate within regulated timelines. Ideal for transportation, manufacturing, healthcare, construction, energy, government, and any industry with mandated or policy-driven drug testing programs.
The drug screening process typically involves the HR or compliance team who initiates the test order, the employee or candidate who reports for collection, the collection site staff who collect the specimen under chain-of-custody protocols, the laboratory that analyzes the specimen, the medical review officer (MRO) who reviews and verifies results, and the designated employer representative (DER) who receives the final determination.
Compliant test administration by ensuring every step — from ordering through result delivery — follows regulated chain-of-custody and documentation requirements. Faster turnaround from test order to final result by routing each phase to the right participant with clear instructions and deadlines. Confidential result handling with access controls that ensure only authorized parties view test results and MRO determinations. Reduced testing errors and disputes through structured collection procedures, specimen tracking, and MRO review before results are finalized. Complete compliance documentation for every test administered, supporting audit, litigation defense, and regulatory reporting requirements.
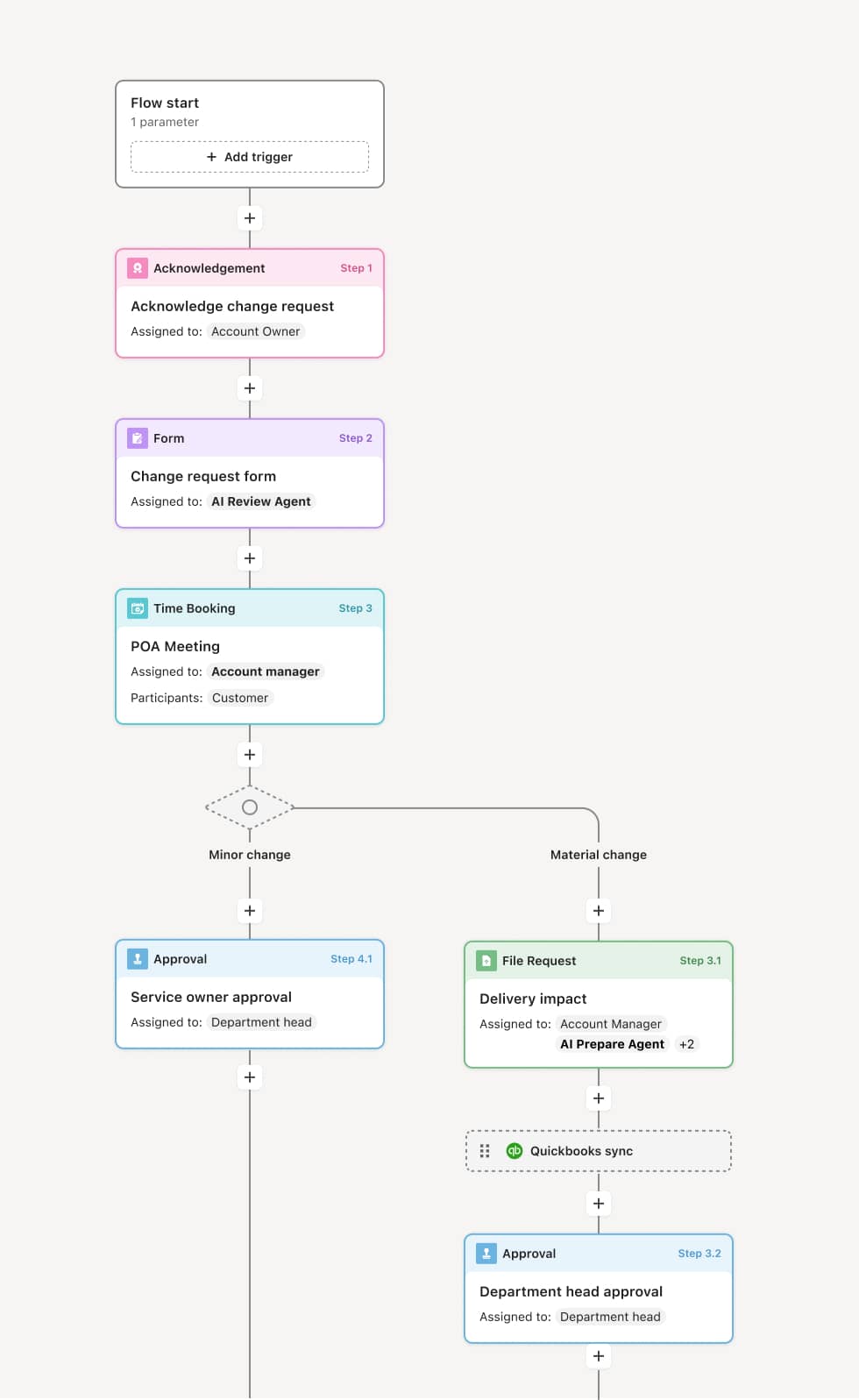
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo’s flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Test order initiation
The process begins when HR or the compliance team generates a drug test order based on the applicable testing reason — pre-employment, random selection, post-incident, reasonable suspicion, or return-to-duty. The order includes the testing panel, collection site assignment, and a deadline for the individual to report for collection. An AI Agent can assist by selecting the appropriate testing panel based on the reason for testing and applicable regulatory requirements.
Specimen collection
The designated individual reports to the assigned collection site within the required timeframe. Collection staff perform the specimen collection following chain-of-custody protocols, including identity verification, specimen labeling, tamper-evident sealing, and proper documentation. The chain-of-custody form accompanies the specimen to the laboratory.
Laboratory analysis
The laboratory receives the specimen, verifies chain-of-custody integrity, and performs the screening analysis. If the initial screen is negative, the result is reported. If the screen is positive or inconclusive, a confirmation test is performed. The laboratory reports the result to the medical review officer.
Medical review officer (MRO) review
The MRO reviews the laboratory result. For positive or inconclusive results, the MRO contacts the individual to determine if a legitimate medical explanation exists. Based on this review, the MRO issues a final verified result — negative, positive, or other determination. An AI Agent may prepare the MRO’s review package with the test order details, laboratory results, and any relevant prior testing history.
Result delivery and determination
The final verified result is communicated to the designated employer representative (DER) through a secure, confidential channel. If the result is negative, the individual is cleared. If the result is positive, the HR or compliance team initiates the appropriate next steps based on policy — which may include follow-up assessment, disciplinary action, or return-to-duty planning.
Documentation and record retention
The complete drug screening record — including the test order, chain-of-custody documentation, laboratory report, MRO determination, and employer action — is preserved in compliance with applicable retention requirements.
This process commonly relies on inputs such as the test order, individual identification, chain-of-custody forms, laboratory analysis results, and MRO review documentation. It may be triggered by a hiring event, random selection, an incident report, a supervisor’s reasonable suspicion determination, or a return-to-duty requirement. Connected systems often include occupational health platforms, HRIS systems like Workday or ADP for employee data, laboratory information systems for specimen tracking, and compliance management tools for regulatory reporting.
Key decision points include the selection of the appropriate testing panel and collection site based on the reason for testing, whether the individual reports for collection within the required timeframe, whether the laboratory result requires confirmation testing, whether the MRO determines a legitimate medical explanation for a positive result, and what employer action is taken based on the final verified result.
Individuals not reporting for collection within the required window, creating compliance gaps and requiring follow-up or policy enforcement. Chain-of-custody errors at the collection site that compromise specimen integrity and result defensibility. Delayed MRO review when the officer is not available to contact the individual promptly for positive result verification. Result confidentiality breaches when test results are shared with unauthorized parties or through unsecured channels. Incomplete record retention that leaves the organization unable to demonstrate compliance during audits or litigation.
Orchestrates the full drug screening lifecycle from test order through result delivery across HR, collection sites, laboratories, and the MRO in a single secure flow.
Routes each phase to the right participant with clear instructions, deadlines, and documentation requirements to maintain compliance.
AI Agents select the appropriate testing panel and flag regulatory requirements based on the reason for testing, reducing order errors.
Maintains confidentiality controls so test results are visible only to authorized parties at each stage of the process.
Connects to HRIS and occupational health systems like Workday, ADP, and compliance platforms so testing data and employee records stay synchronized.
Preserves the complete screening record including test order, chain-of-custody documentation, laboratory results, MRO determination, and employer actions for regulatory compliance and audit readiness.