Engineering change coordinator
Configuration manager
Chief engineer
Manufacturing engineering manager
Quality director
R&D director

When product designs require formal modification. When manufacturing processes need specification updates. When quality issues drive design corrections. When cost reduction requires engineering changes. Ideal for manufacturing, aerospace, automotive, medical devices, and any regulated or engineering-intensive industry.
Engineers initiate change requests with technical justification. Change coordinators manage the review process. Affected departments evaluate impacts within their domains. Change review boards evaluate significant changes. Configuration management maintains change records.
Controlled engineering changes with formal authorization process Complete impact assessment across all affected functions Regulatory compliance with documented change history Coordinated implementation across manufacturing, quality, and supply chain
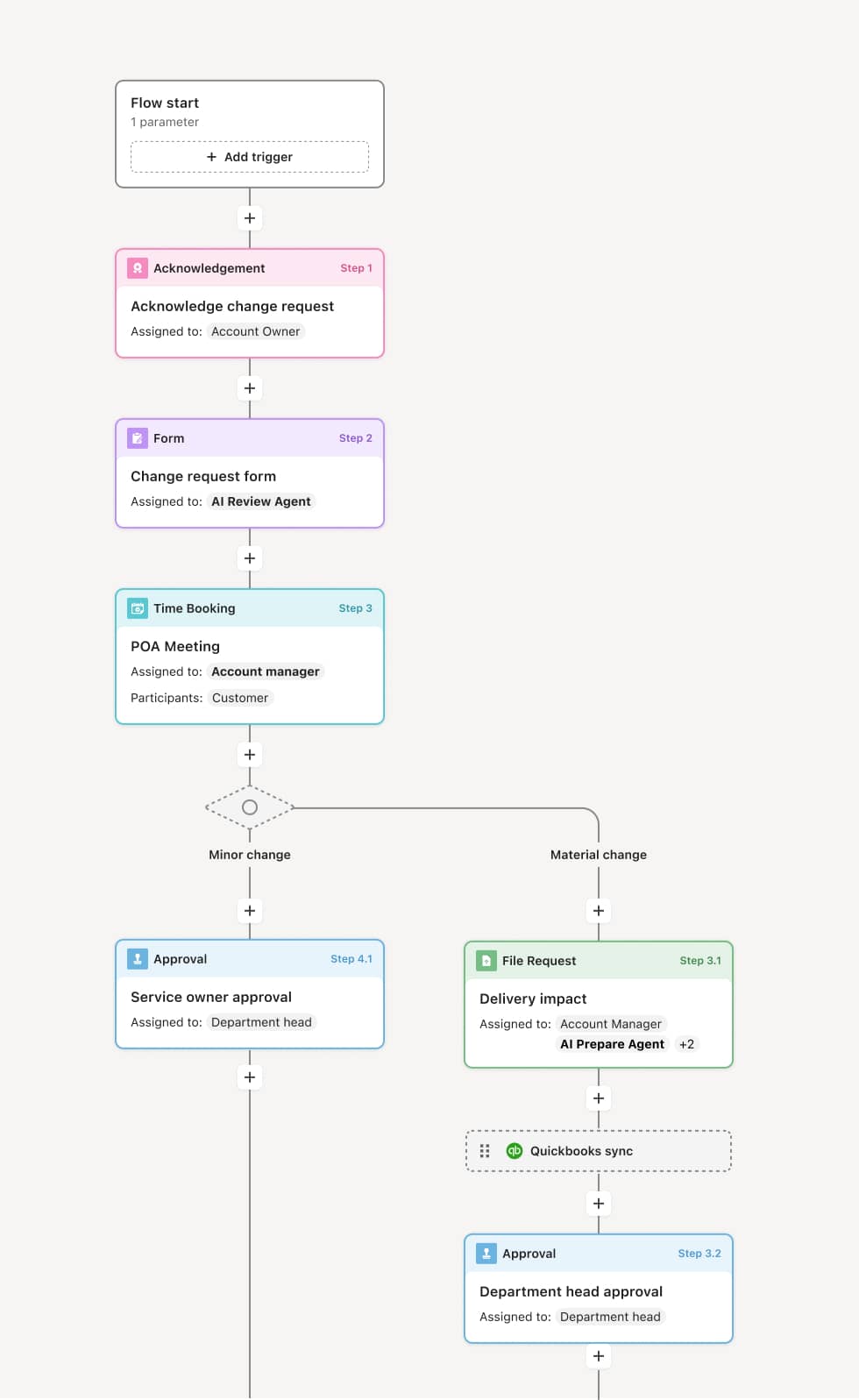
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo's flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Change request initiation
The process begins when an engineering change request is submitted including the proposed modification, affected items, technical justification, and preliminary impact assessment. AI agents may identify all affected documents and components automatically.
Impact analysis
Each affected function analyzes impacts within their domain. Manufacturing assesses production implications. Quality evaluates testing and validation needs. Supply chain reviews inventory and supplier impacts. Costs and timelines are estimated.
Technical review
Engineering reviews the change for technical soundness and completeness. They verify that the modification achieves objectives without introducing new problems. Design verification requirements are established.
Change board authorization
Significant changes are presented to the change control board for authorization. The board reviews impact analyses, costs, and implementation plans. They approve, modify, or reject the change with documented rationale.
Implementation coordination
Approved changes are coordinated for implementation with defined effectivity. Document updates, inventory disposition, and supplier notifications are managed. Change completion is verified and recorded.
This process relies on engineering documents, bills of materials, impact assessments, and change requests. Triggers include design issues, quality findings, cost initiatives, or regulatory changes. Integration with PLM systems like Siemens Teamcenter or Arena, ERP systems, and document management supports change control.
Key decision points include determining whether the change is necessary and technically sound, whether all impacts have been identified, whether costs and benefits are acceptable, and whether implementation timing is appropriate.
Changes approved without complete impact analysis causing surprises. Affected documents not updated creating configuration errors. Inventory disposition not managed causing obsolete stock. Change history incomplete affecting regulatory audits.
Orchestrates engineering change review across all affected functions with structured impact analysis
AI agents identify affected items and documents automatically
Coordinates change board review and authorization
Maintains complete change history for configuration management and compliance