Laboratory operations manager
Specimen processing supervisor
Logistics coordinator
Quality assurance manager
Clinical operations director
Compliance officer

This process is used when collected specimens must be tracked through transport, receipt, accessioning, analysis, and result reporting to maintain chain-of-custody documentation and ensure timely processing. It applies when specimens cross organizational boundaries — from collection sites to reference laboratories, between departments, or through courier services — and when handling conditions such as temperature, timing, and transport medium must be monitored. It is common when collection staff, couriers, laboratory receiving, and processing teams must coordinate to prevent specimen loss, mishandling, or delays. Ideal for hospitals, reference laboratories, clinical research organizations, public health agencies, and any entity handling diagnostic or research specimens at scale.
The specimen tracking process typically involves collection site staff who label and package specimens, courier or transport teams who move specimens to the laboratory, laboratory receiving staff who accession and verify specimen integrity, laboratory technicians who process and analyze specimens, and quality assurance teams who monitor handling compliance and investigate discrepancies.
Real-time specimen visibility so every stakeholder knows where a specimen is, who handled it, and whether it is on schedule for processing. Fewer lost or misrouted specimens because every handoff is documented and verified within the tracking workflow. Preserved specimen integrity through monitoring of handling conditions, transport time, and processing windows at each stage. Faster issue resolution when specimen discrepancies are detected because the chain-of-custody record identifies exactly where the breakdown occurred. Compliance-ready documentation that satisfies regulatory and accreditation requirements for specimen handling and custody.
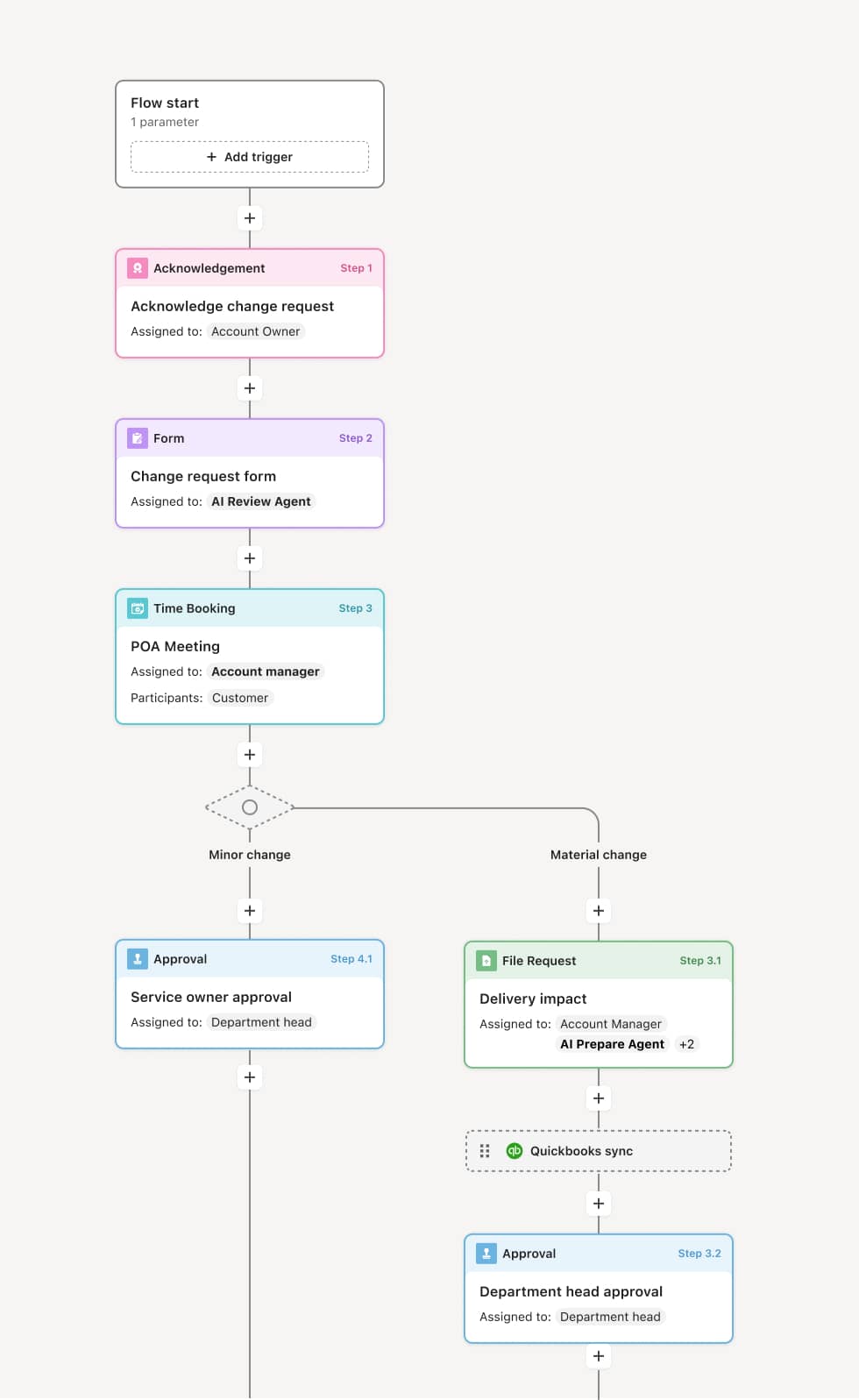
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo’s flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Specimen collection and labeling
The process begins when a specimen is collected and labeled with patient identification, test order details, collection time, and handling requirements. The collection record is entered into the tracking system. An AI Agent can assist by verifying that the label matches the test order and that handling instructions are complete before the specimen leaves the collection site.
Packaging and transport handoff
The specimen is packaged according to transport requirements — including temperature control, biohazard containment, and timing constraints. The handoff to the courier or transport team is documented with a timestamp and condition verification. If multiple specimens are batched for transport, the manifest is logged.
Transport monitoring
During transport, the specimen’s status is tracked. For temperature-sensitive specimens, condition monitoring may be active. If transport exceeds acceptable time windows or conditions deviate from requirements, an alert is generated for the receiving laboratory. An AI Agent may flag specimens approaching their processing deadline during transit.
Laboratory receiving and accessioning
Upon arrival, laboratory receiving staff verify the specimen against the manifest, confirm label integrity, and assess specimen condition. If a specimen is damaged, mislabeled, or does not meet acceptance criteria, it is flagged for rejection or recollection and the ordering provider is notified. Accepted specimens are accessioned into the laboratory information system.
Processing and analysis
The specimen enters the laboratory workflow for the ordered analysis. Processing status is tracked through each analytical phase. If processing issues arise — such as insufficient volume or quality concerns — the specimen may be flagged for repeat testing or recollection.
Result delivery and record closure
Upon completion of analysis, results are delivered to the ordering provider. The specimen tracking record is closed with the complete chain-of-custody documentation, processing timestamps, and any exception notes preserved for regulatory compliance and quality review.
This process commonly relies on inputs such as specimen labels, collection records, transport manifests, condition monitoring data, and laboratory accessioning records. It may be triggered by a specimen collection event, a transport dispatch, or a laboratory receiving action. Connected systems often include laboratory information systems (LIS) like Sunquest or Beaker, specimen tracking platforms, courier management systems, and EHR platforms like Epic or Cerner for order data.
Key decision points include whether the specimen meets labeling and packaging requirements before leaving the collection site, whether transport conditions remain within acceptable parameters during transit, whether the specimen passes receiving acceptance criteria at the laboratory, and whether processing issues require recollection or re-testing.
Labeling errors at collection that create mismatches between the specimen and the test order, requiring recollection and delaying results. Transport condition breaches when temperature or timing requirements are not maintained, compromising specimen integrity. Receiving discrepancies when specimens arrive without matching manifests or with damaged packaging, stalling accessioning. Processing bottlenecks that cause specimens to exceed stability windows before analysis. Incomplete chain-of-custody records that cannot demonstrate handling compliance during audits or investigations.
Orchestrates specimen tracking from collection through result delivery across collection sites, transport, laboratory receiving, and processing teams in a single coordinated flow.
Documents every handoff with timestamps, condition verification, and responsible party identification to maintain chain-of-custody integrity.
AI Agents verify specimen labeling and completeness at collection and flag handling requirement gaps before the specimen enters transport.
Monitors processing timelines and alerts stakeholders when specimens approach stability or processing deadlines.
Connects to LIS and EHR platforms like Sunquest, Beaker, and Epic so specimen data, orders, and results stay synchronized across systems.
Preserves the complete chain-of-custody record for every specimen, including collection, transport, receiving, processing, and exception documentation for regulatory compliance and quality assurance.