Regulatory affairs manager
Compliance director
Legal counsel
Operations director
Quality assurance manager
Regulatory submissions specialist

This process is used when a product launch, facility modification, clinical trial, service offering, or business activity requires formal approval from a regulatory authority before it can proceed. It is triggered when a new submission is required, when a renewal or amendment is due, or when a regulatory inquiry or deficiency notice must be addressed. Regulatory approval applies when submissions involve extensive documentation, cross-functional input, and defined timelines for agency response. It is common in pharmaceuticals, medical devices, financial services, energy, telecommunications, food production, and any industry subject to government oversight.
The regulatory affairs team leads the submission process, coordinating content preparation and agency communication. Legal reviews the submission for legal sufficiency and risk. Quality assurance contributes technical documentation and validation data. Operations provides operational context and implementation plans. External consultants or regulatory agents may be engaged for specialized submissions. Executive leadership may be involved for submissions with significant business impact or strategic importance.
On-time submissions because the preparation process is structured to ensure all required documentation, data, and reviews are completed before filing deadlines. Reduced deficiency responses through comprehensive pre-submission review that catches gaps, inconsistencies, and missing data before the agency identifies them. Clear correspondence tracking so that every agency communication, deficiency notice, and response is captured and accessible to the full regulatory team. Faster time to approval by coordinating cross-functional input in parallel where possible, rather than sequentially assembling the submission package. Auditable submission history with a complete record of every document, review, agency interaction, and approval decision for compliance and governance purposes.
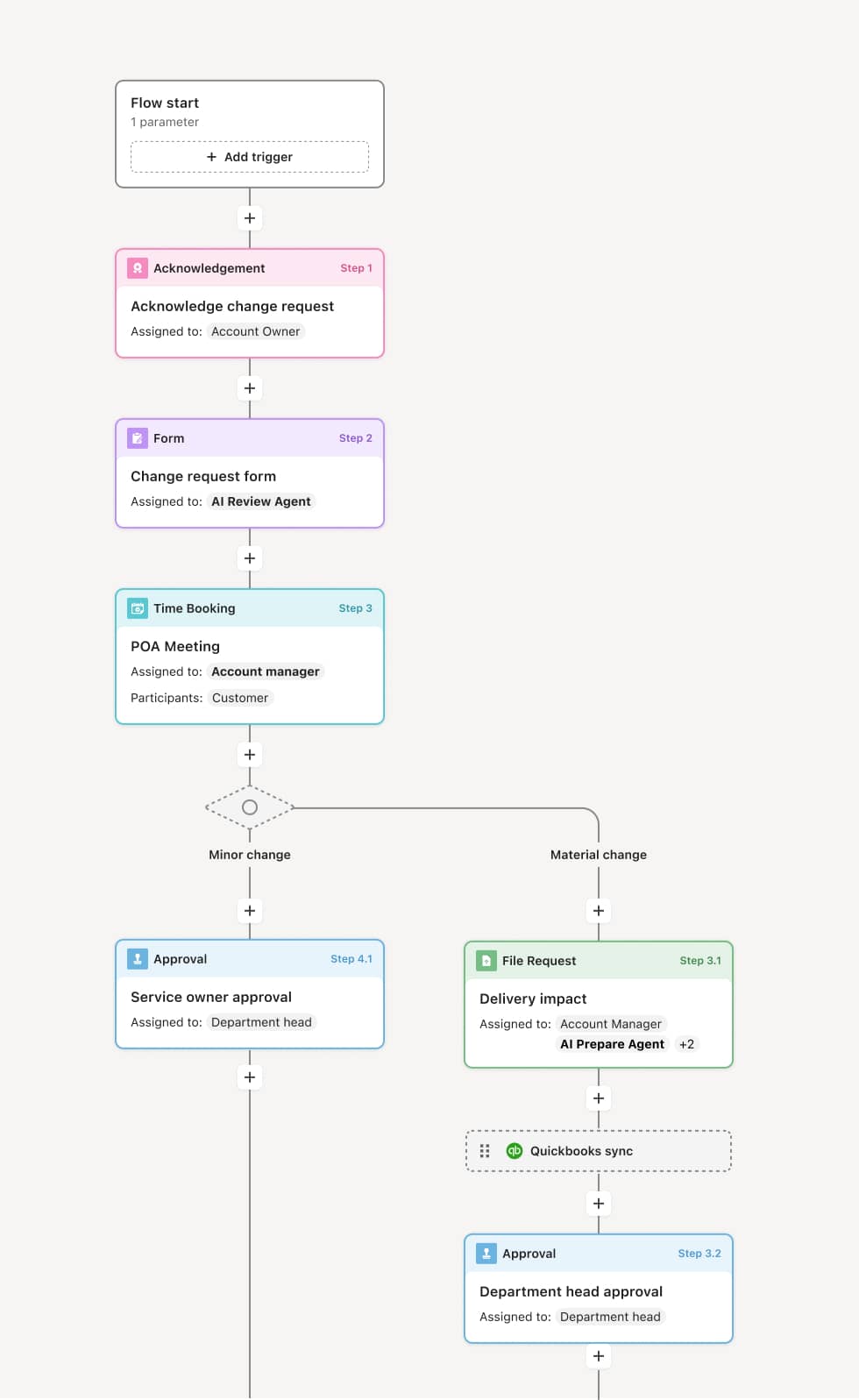
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo’s flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Submission planning and requirements mapping
The process begins when a regulatory submission is identified as required. The regulatory affairs team defines the submission type, applicable regulations, required documentation, agency-specific formatting requirements, and the target filing date. An AI agent can assist by pulling relevant regulatory guidance, identifying required document types based on the submission category, and creating a preparation checklist that maps each requirement to a responsible contributor.
Cross-functional content preparation
The required documentation is assembled from across the organization. Quality provides technical data, validation reports, and testing results. Operations contributes process descriptions and implementation plans. Legal reviews risk disclosures and regulatory language. Each contributor prepares their section within defined timelines, and the regulatory affairs team coordinates the assembly. If any content is incomplete or does not meet agency formatting standards, it is returned to the contributor with specific feedback. AI agents can validate document completeness and flag formatting issues before the package is compiled.
Pre-submission review and quality check
Before the submission is filed, the complete package undergoes a final review. This includes checking for consistency across documents, verifying that all required sections are present, confirming that data references are accurate, and ensuring that the submission meets the agency’s current requirements. If any issues are identified, the process loops back to the relevant contributor for correction. For high-risk submissions, an internal review committee may conduct a formal readiness assessment.
Filing and agency correspondence management
The submission is filed with the regulatory authority through the required channel. Once submitted, the process shifts to tracking agency correspondence—including acknowledgments, requests for additional information, deficiency notices, and interim communications. Each correspondence item is captured within the workflow and routed to the appropriate team for response. If the agency requests additional data or raises questions, the process coordinates the response across the necessary contributors with defined timelines.
Approval, conditions, and post-approval implementation
When the regulatory authority issues a decision, the outcome is recorded—whether approval, conditional approval, or denial. If conditions are attached, they are tracked as action items assigned to the responsible teams. Post-approval implementation steps—such as labeling changes, manufacturing updates, or compliance monitoring requirements—are initiated within the same workflow. The complete submission record, including all correspondence and the final decision, is retained for regulatory compliance and future reference.
This process commonly relies on inputs such as regulatory guidance documents, product or service specifications, technical and validation data, risk assessments, legal opinions, and prior submission records. It may be triggered by a product development milestone, a regulatory calendar event, a change control decision, or an agency inquiry. Connected systems such as Veeva Vault, MasterControl, or SharePoint may manage document repositories, while ERP systems like SAP or Oracle provide product and manufacturing data.
Key decision points include whether all required documentation meets agency standards before filing, whether the pre-submission review identifies any gaps that must be resolved, how to respond to agency deficiency notices or requests for additional information, and whether conditions attached to an approval can be met within required timelines. If the submission is denied, the process branches to determine whether a revised submission, appeal, or alternative approach is appropriate.
Submission packages filed with missing or incomplete documentation, resulting in agency deficiency notices that delay the approval timeline. Cross-functional contributors not engaged early enough, causing last-minute rushes that compromise submission quality. Agency correspondence not tracked centrally, leading to missed response deadlines or inconsistent communication. Pre-submission review skipped or superficial, allowing preventable errors to reach the agency. Post-approval conditions not tracked to completion, creating compliance gaps after the initial approval is granted.
Orchestrates regulatory submissions across compliance, legal, quality, operations, and external consultants so that every contributor is engaged at the right time with clear responsibilities and deadlines.
AI agents assist with submission readiness by validating document completeness, checking formatting against agency requirements, and flagging gaps before the package is filed.
Tracks agency correspondence within the same workflow, ensuring that deficiency notices, requests for information, and interim communications are captured and routed to the appropriate responders with defined timelines.
Connects to document management and regulatory systems such as Veeva Vault, MasterControl, or SharePoint to assemble submission packages from existing repositories without manual document gathering.
Maintains a complete, auditable record of every submission, correspondence, response, and regulatory decision, supporting compliance governance and providing a reference foundation for future submissions.