Clinical research associate
Clinical operations manager
Principal investigator
Clinical research coordinator
Quality assurance auditor
Sponsor study director

This process is used when a sponsor or CRO conducts a routine, interim, or for-cause monitoring visit at a clinical trial site to verify that the trial is being conducted according to the protocol, that subject rights and safety are protected, that reported data is accurate and complete, and that the site is maintaining regulatory documentation. Monitoring visits may be conducted on-site, remotely, or through a hybrid approach depending on the trial and regulatory expectations. Ideal for pharmaceutical sponsors, CROs, and clinical trial sites managing site monitoring under FDA, EMA, or ICH-GCP oversight.
The monitoring visit process typically involves clinical research associates (CRAs) who plan and conduct the visit, clinical research coordinators who prepare site documentation and assist during the visit, principal investigators who discuss study conduct and findings, sponsor clinical operations managers who oversee monitoring activities, and quality assurance staff who review monitoring reports for trends.
Verified protocol compliance through systematic review of site conduct, subject eligibility, informed consent, and data integrity. Timely identification of site issues so corrective actions can be implemented before problems affect data quality or subject safety. Accurate and complete data confirmed through source data verification against case report forms. Documented regulatory readiness with complete monitoring reports and site files that support inspection. Efficient visit execution because pre-visit planning, on-site activities, and follow-up are managed in a coordinated workflow.
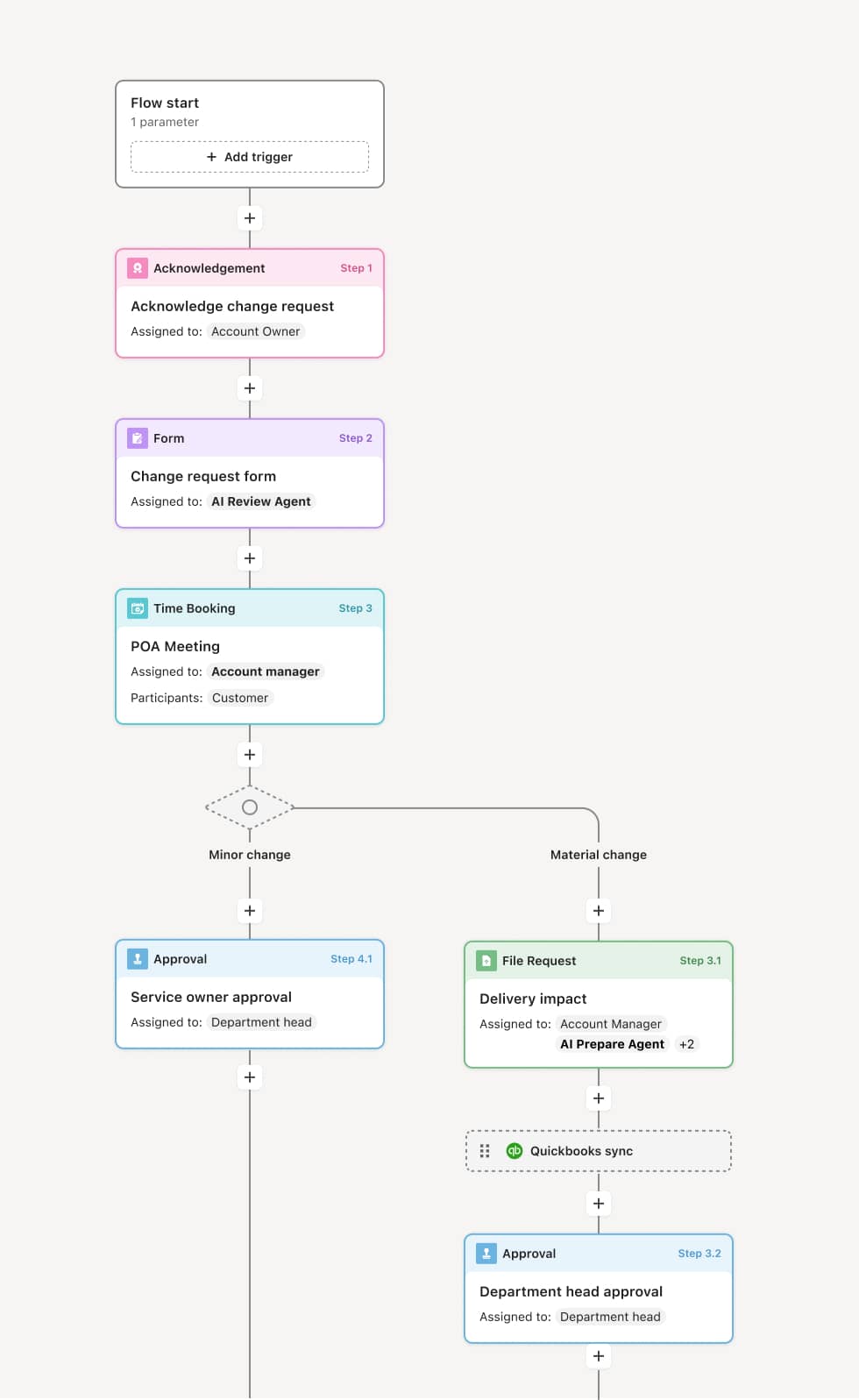
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo’s flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Pre-visit planning and preparation
The process begins when the CRA plans the monitoring visit, identifying the subjects, documents, and activities to be reviewed. The CRA reviews prior monitoring reports, open action items, enrollment status, and data queries. An AI Agent can assist by compiling open findings from previous visits, flagging subjects with outstanding queries, and generating the visit agenda based on the monitoring plan.
Site notification and document preparation
The site is notified of the visit dates and the specific documents and subjects to be reviewed. The clinical research coordinator prepares the site files, subject binders, regulatory documents, and source records for the CRA’s review.
On-site monitoring activities
The CRA conducts the visit, which typically includes source data verification (comparing CRF data against source records), informed consent review, regulatory document review, drug accountability, adverse event reconciliation, and protocol compliance assessment. Findings are documented during the visit.
Monitoring report and findings documentation
After the visit, the CRA prepares the monitoring visit report documenting all activities performed, findings identified, and the site’s overall compliance status. Findings are classified by severity and assigned follow-up actions with responsible parties and due dates. An AI Agent may compare findings against common categories and prior visit patterns to identify recurring issues.
Follow-up action tracking
Open findings and corrective actions are assigned to the site and tracked through resolution. The CRA verifies that corrective actions are implemented at the next visit or through remote follow-up. Overdue actions are escalated to the sponsor.
Report finalization and filing
The monitoring report is finalized, reviewed by the sponsor, and filed in the trial master file. The visit record, including findings, corrective actions, and resolution documentation, is preserved for regulatory inspection.
This process commonly relies on inputs such as the monitoring plan, prior monitoring reports, open action items, subject enrollment data, CRF and source data, regulatory binder documents, and drug accountability records. It may be triggered by the monitoring schedule in the study plan. Connected systems often include CTMS platforms like Medidata or Veeva Vault, EDC systems for CRF data, and trial master file systems for document management.
Key decision points include which subjects, documents, and activities to prioritize for review during the visit, how findings are classified by severity and what corrective actions are assigned, whether recurring findings indicate a systemic site issue requiring escalation, and whether the site’s overall compliance status warrants changes to the monitoring frequency or approach.
Pre-visit planning inadequate, resulting in inefficient use of on-site time and missed review priorities. Site not prepared with required documents and source records, delaying the CRA’s review activities. Findings documented without clear corrective action assignments, making follow-up ambiguous. Follow-up actions not tracked to resolution, allowing the same findings to recur at subsequent visits. Monitoring reports delayed after the visit, creating gaps in the trial master file and reducing the value of timely findings.
Orchestrates the monitoring visit lifecycle from pre-visit planning through follow-up resolution across CRAs, site staff, and sponsors in a single coordinated flow.
AI Agents compile open findings, subject queries, and review priorities to prepare the CRA’s visit agenda based on the monitoring plan.
Engages site staff within the workflow for visit notification, document preparation requests, and finding response and corrective action tracking.
Tracks corrective action implementation for every finding, escalating overdue actions and verifying resolution.
Connects to CTMS and EDC platforms like Medidata and Veeva Vault so monitoring data, subject records, and trial master file documents are accessible in context.
Preserves the complete monitoring visit record including the visit report, findings, corrective actions, and resolution documentation for regulatory inspection and quality oversight.