Clinical operations manager
Site activation lead
Regulatory affairs coordinator
Principal investigator
Clinical research coordinator
Study start-up specialist

This process is used after a site has been selected for a clinical trial and must complete all regulatory, contractual, operational, and training requirements before being authorized to screen and enroll subjects. It applies when regulatory submissions (IRB/EC), site agreements, investigator qualifications, system access, drug supply, and training must be coordinated across the sponsor, CRO, and site staff within the study timeline. It is common when site activation timelines directly affect enrollment targets and overall study milestones. Ideal for pharmaceutical sponsors, CROs, academic medical centers, and clinical trial sites managing study start-up activities.
The site onboarding process typically involves site activation leads who manage the overall onboarding timeline, regulatory coordinators who prepare and track regulatory submissions, principal investigators and site staff who complete training and regulatory documentation, contracts and budget teams who negotiate and execute site agreements, and clinical operations managers who authorize site activation upon readiness confirmation.
Faster site activation by managing all onboarding activities in parallel rather than waiting for sequential completion. Complete regulatory readiness with all IRB/EC approvals, regulatory documents, and essential documents in place before enrollment begins. Trained site staff who are prepared to conduct the study according to the protocol and GCP requirements. Documented activation milestones that support inspection readiness from the first day of enrollment. Reduced enrollment delays because sites are activated on schedule and ready to screen subjects as planned.
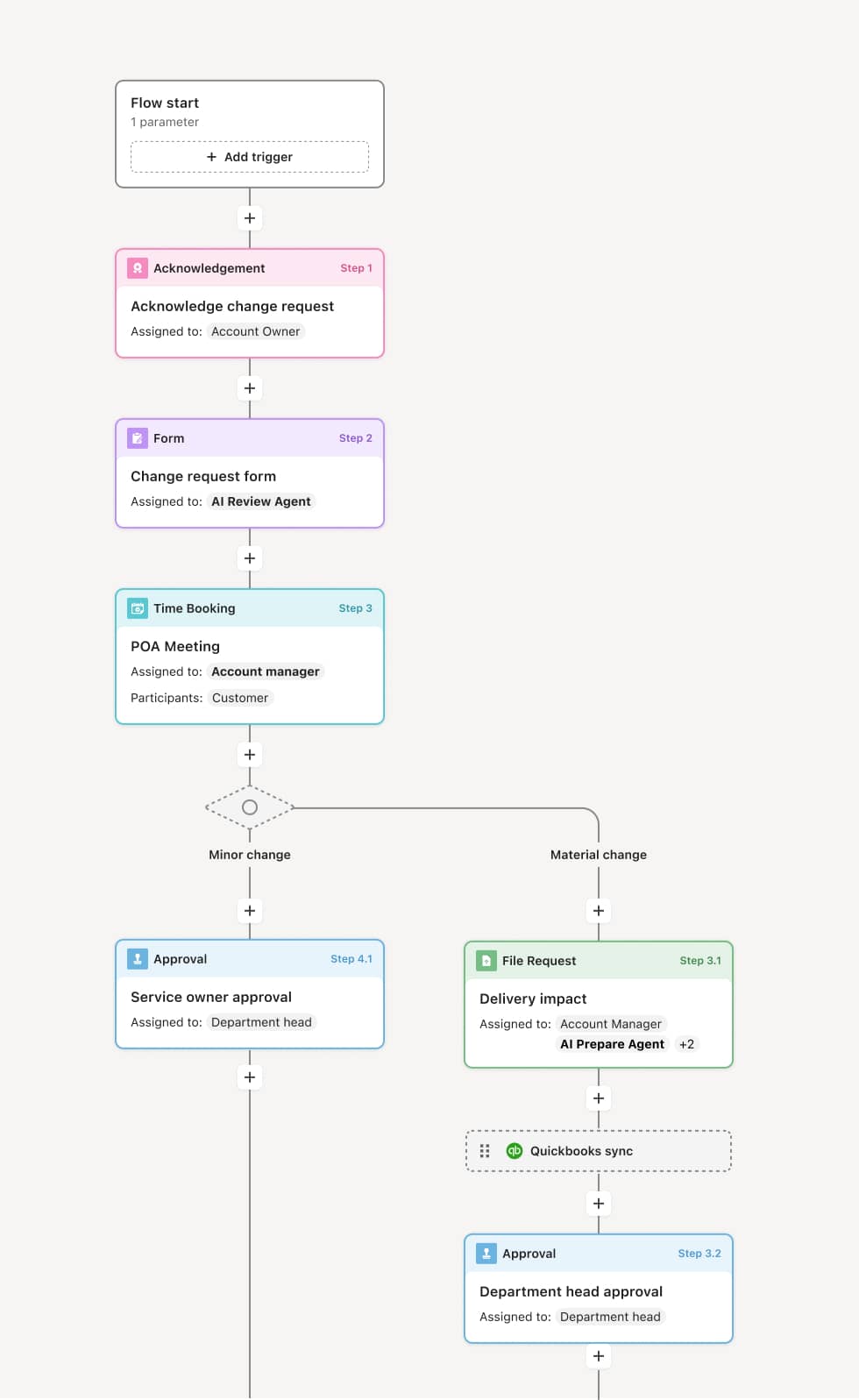
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo’s flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Onboarding initiation and milestone planning
The process begins when a site is selected and the activation lead creates the onboarding plan, identifying all required milestones: regulatory submission, agreement execution, training completion, system provisioning, drug supply, and readiness confirmation. An AI Agent can assist by generating the milestone checklist based on the study protocol, country-specific requirements, and the site’s prior trial experience.
Regulatory document collection and submission
The regulatory coordinator works with the site to collect and prepare required regulatory documents — including the investigator’s brochure acknowledgment, CVs, medical licenses, financial disclosures, IRB/EC submission package, and protocol signature pages. The IRB/EC submission is tracked through approval. An AI Agent may verify document completeness and flag missing or expired items before submission.
Contract and budget negotiation
The contracts team and the site negotiate the clinical trial agreement and budget. Terms are reviewed by legal, finance, and the site, with revisions tracked through execution. The fully executed agreement is filed in the trial master file.
Investigator and staff training
Site staff complete required training on the study protocol, GCP, EDC system, study-specific procedures, and safety reporting. Training completion is documented with certificates or attestation records. The sponsor confirms that all required staff are trained before activation.
System access and supply provisioning
EDC, IVRS/IWRS, and other study system accounts are provisioned for trained site staff. Drug supply and clinical supplies are shipped and confirmed received at the site. An AI Agent may track system access provisioning and supply delivery status across all required systems.
Site readiness confirmation and activation
The activation lead reviews all milestones — regulatory approval, executed agreement, completed training, system access, and supply receipt — and confirms that the site is ready to begin screening. The sponsor authorizes activation and the site is cleared to enroll subjects.
This process commonly relies on inputs such as the study protocol, regulatory document packages, site CVs and licenses, clinical trial agreements, training materials, and system access requests. It may be triggered by site selection and feasibility completion. Connected systems often include CTMS platforms like Medidata or Veeva Vault, regulatory tracking systems, EDC platforms, and contract management systems.
Key decision points include whether all regulatory documents are complete and the IRB/EC submission is ready, whether contract and budget terms are agreed and the agreement is executed, whether all required site staff have completed training, and whether all activation milestones are confirmed before the site is authorized to enroll.
Regulatory document collection delays when site staff do not provide required documents in a timely manner. IRB/EC review timelines longer than expected, pushing back the activation date. Contract negotiations stalled because budget or liability terms require multiple revision cycles. Training not completed for all required staff before the activation deadline. System access not provisioned in time for the site to begin data entry at enrollment start.
Orchestrates site onboarding across regulatory, contracts, training, systems, and operations in a single workflow that tracks every activation milestone to completion.
Engages site staff directly within the workflow for document submission, training completion, and readiness confirmation, keeping all interactions in context.
AI Agents generate milestone checklists based on the study and country requirements and track completion status across all parallel workstreams.
Manages parallel onboarding tracks so regulatory, contracts, training, and system provisioning proceed concurrently rather than sequentially.
Connects to CTMS, regulatory tracking, and EDC platforms like Medidata and Veeva Vault so onboarding data and activation status are synchronized.
Preserves the complete onboarding record including regulatory approvals, executed agreements, training documentation, and activation confirmation for trial master file and inspection readiness.