Principal investigator
IRB coordinator
Research compliance officer
Institutional review board chair
Clinical research manager
Regulatory affairs specialist

This process is used when a research study involving human subjects requires institutional ethics review and approval before data collection or participant enrollment can begin. It applies when new protocols, amendments to existing protocols, or continuing review submissions must be evaluated against ethical guidelines such as the Common Rule, ICH-GCP, or institutional policies. It is common when multiple board members, compliance officers, and external reviewers must coordinate their assessments within regulatory timelines. Ideal for academic medical centers, contract research organizations, pharmaceutical companies, and any institution conducting human subjects research.
The IRB approval process typically involves principal investigators who submit protocols and respond to board queries, IRB coordinators who manage submissions and track review progress, IRB board members and chairs who conduct ethical and scientific review, research compliance officers who verify regulatory alignment, and occasionally external reviewers or community members who provide additional perspectives on participant risk and benefit.
Shorter time to IRB authorization by routing submissions to the correct review pathway — expedited, exempt, or full board — based on risk classification. Fewer incomplete submissions through upfront validation of required documents, consent forms, and protocol elements before board review begins. Clear regulatory traceability with documented review decisions, stipulations, and approvals that satisfy audit and accreditation requirements. Reduced investigator burden by guiding principal investigators through the submission, response, and resubmission cycle with structured prompts and context. Consistent ethical oversight across studies through standardized review criteria and board-level decision records.
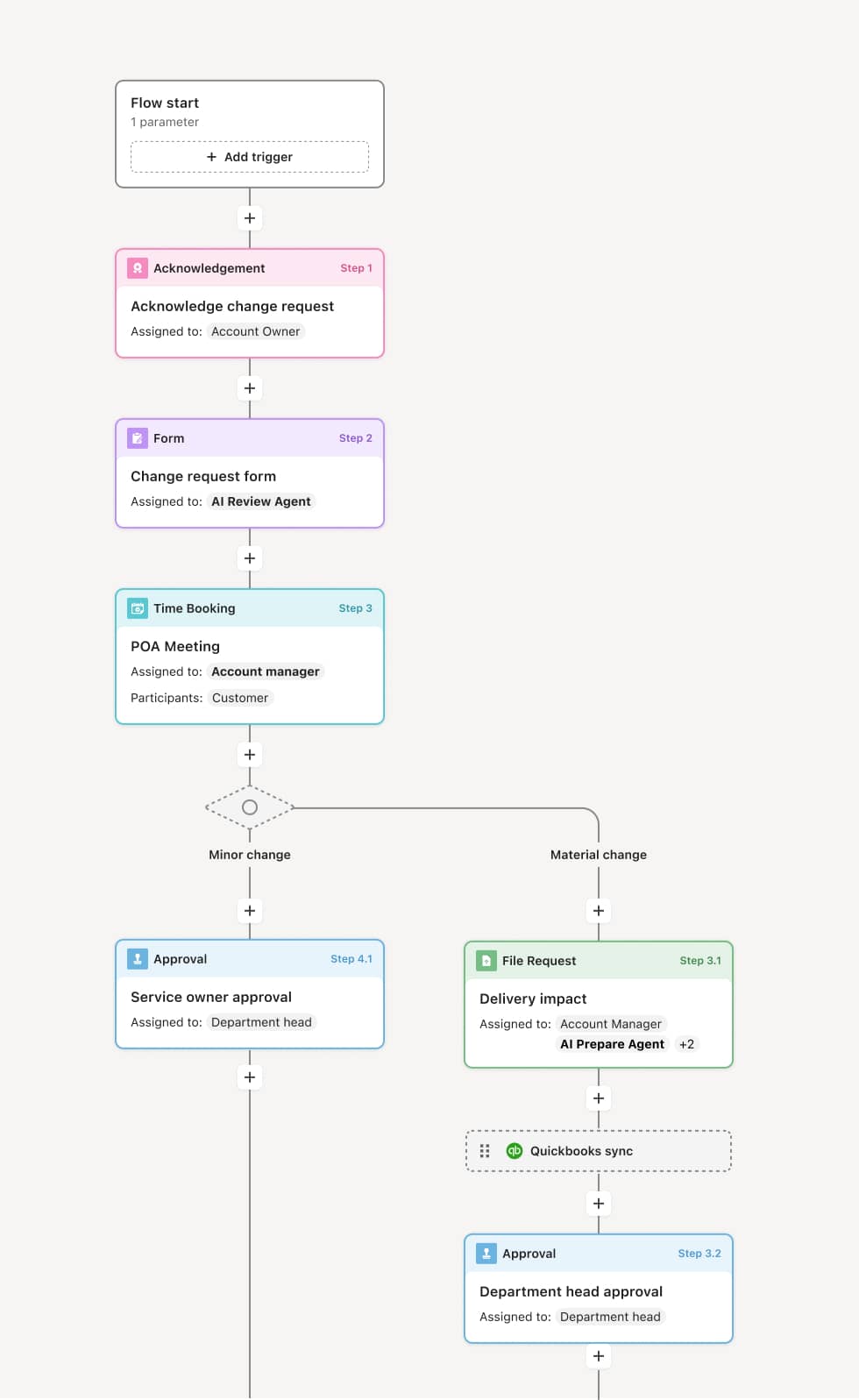
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo’s flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Protocol submission
The process begins when a principal investigator submits a research protocol, informed consent documents, recruitment materials, and any supporting instruments for IRB review. An AI Agent can assist by checking the submission package for completeness — verifying that required sections such as risk assessment, participant protections, and data management plans are included before routing to the IRB office.
Administrative pre-review and classification
The IRB coordinator reviews the submission for administrative completeness and classifies the study by risk level. Exempt, expedited, and full board review pathways each carry different timelines and reviewer requirements. If the submission is incomplete or misclassified, it is returned to the investigator with specific guidance.
Reviewer assignment and scientific review
Based on the classification, the submission is assigned to one or more board reviewers. For full board review, primary and secondary reviewers conduct detailed assessments of scientific merit, risk-benefit ratio, informed consent adequacy, and participant protections. An AI Agent may prepare a summary of the protocol’s key elements and prior related submissions to support reviewer preparation.
Board deliberation and decision
For full board protocols, the review proceeds to a convened meeting where board members discuss the submission, pose questions to the investigator if needed, and reach a decision. Possible outcomes include approval, approval with stipulations, deferral for revision, or disapproval. The decision and any required modifications are formally documented.
Stipulation response and final approval
If stipulations are issued, the investigator addresses each one and resubmits the relevant materials. The IRB coordinator or designated reviewer verifies that stipulations have been adequately resolved. Once all conditions are satisfied, formal IRB approval is issued with an effective date and approval period.
Post-approval record and notification
The approval letter, along with the final approved protocol, consent documents, and all review correspondence, is recorded. The investigator, research team, and relevant compliance offices are notified. The approved study enters the continuing review cycle for periodic reassessment.
This process commonly relies on inputs such as research protocols, informed consent forms, recruitment materials, investigator qualifications, conflict of interest disclosures, and data management plans. It may be triggered by a new protocol submission, an amendment request, or a continuing review deadline. Connected systems often include electronic IRB platforms like iRIS or IRBManager, research compliance databases, and institutional grant management systems such as Cayuse or Workday.
Key decision points include the initial risk classification that determines the review pathway, whether the submission package is complete enough to enter formal review, the board’s assessment of risk-benefit ratio and participant protections, whether stipulations must be resolved before approval is granted, and the determination of the continuing review interval.
Incomplete submissions that require multiple rounds of administrative back-and-forth before entering review, delaying study timelines significantly. Misclassified risk levels that route protocols to the wrong review pathway, creating rework for the IRB and confusion for investigators. Slow stipulation resolution when investigators lack clarity on what specific changes the board requires. Reviewer availability gaps that delay convened board meetings or expedited reviews beyond regulatory timelines.
Orchestrates the full IRB review lifecycle from submission through approval across investigators, coordinators, board members, and compliance offices in a single coordinated flow.
Routes submissions to the correct review pathway based on risk classification, ensuring exempt, expedited, and full board protocols follow appropriate timelines and reviewer assignments.
AI Agents validate submission completeness at intake, flagging missing protocol sections, consent form elements, or required disclosures before the package reaches the board.
Supports stipulation response loops so investigators address specific board requirements and resubmit without losing context or creating version confusion.
Connects to research compliance systems like iRIS, IRBManager, or Cayuse so submission data, reviewer assignments, and approval records remain synchronized.
Preserves a complete regulatory record of every submission, reviewer comment, board decision, and stipulation resolution, supporting accreditation audits and continuing review.