Procurement manager
Supplier quality engineer
Compliance officer
Vendor management lead
Operations director
Finance reviewer

This process is used when a new supplier must be evaluated and approved before they can be added to the organization’s approved supplier list and participate in procurement activities. It applies when supplier capability, quality certifications, financial health, regulatory compliance, and risk profile must be assessed by multiple functions. It is common when procurement, quality assurance, compliance, and finance teams must coordinate their evaluations before authorization. Ideal for manufacturing, healthcare, pharmaceutical, food and beverage, aerospace, and any industry with regulated or quality-sensitive supply chains.
The supplier approval process typically involves the procurement team or category manager who identifies and nominates the supplier, supplier quality engineers who assess manufacturing or service capability, compliance officers who evaluate regulatory and certification requirements, finance reviewers who assess the supplier’s financial stability, and a procurement director or cross-functional review board who provides final authorization.
Reduced supply chain risk by ensuring every supplier is evaluated for quality, compliance, and financial stability before they enter the supply base. Faster supplier onboarding by routing evaluations to the right teams in parallel rather than sequentially. Consistent qualification standards applied across all suppliers regardless of category, region, or nominating team. Documented supplier due diligence that satisfies audit, regulatory, and customer requirements for supply chain governance. Fewer quality and compliance incidents downstream because capability and certification gaps are identified before the supplier begins delivering.
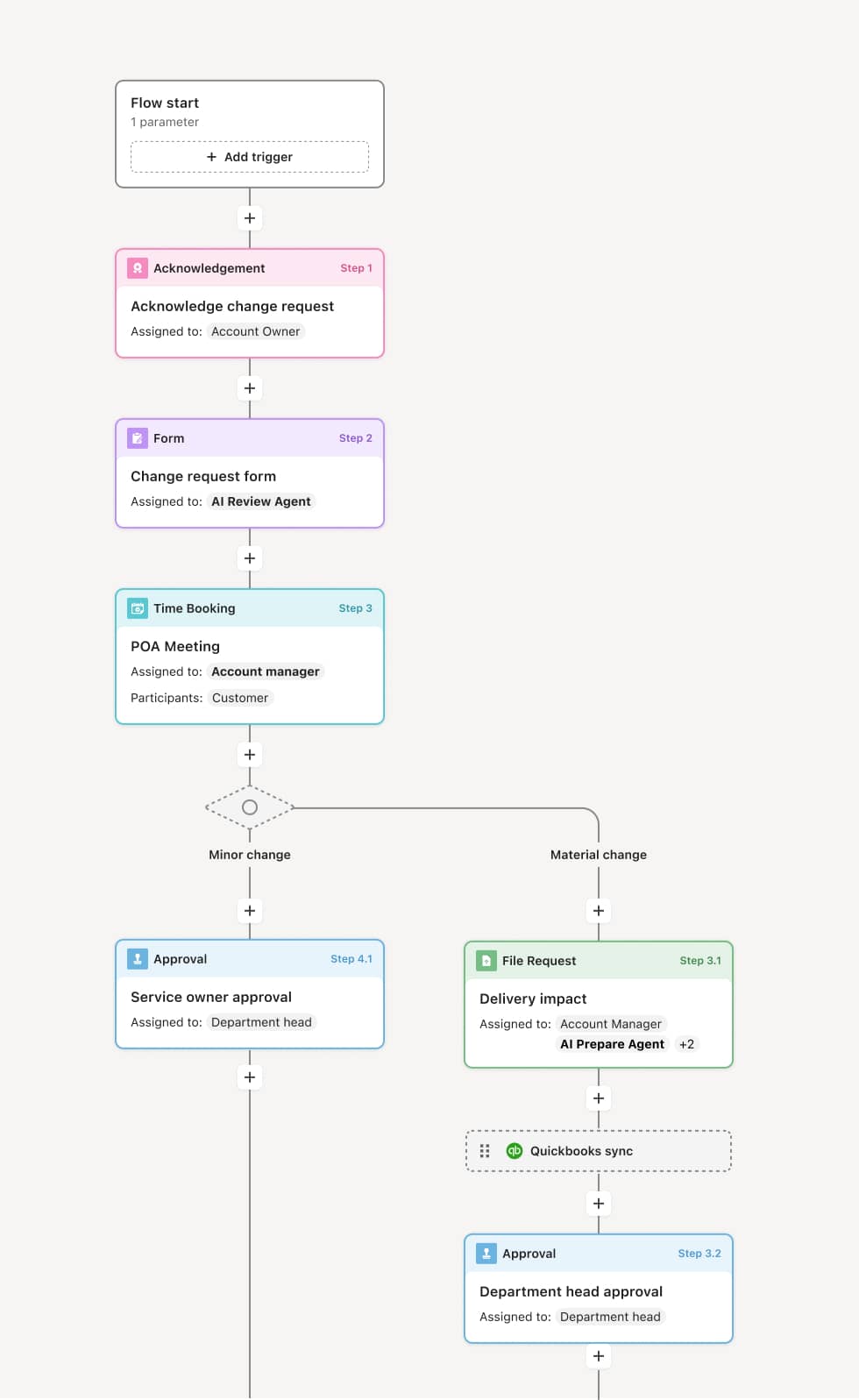
Your version of this process may vary based on roles, systems, data, and approval paths. Moxo’s flow builder can be configured with AI agents, conditional branching, dynamic data references, and sophisticated logic to match how your organization runs this workflow. The steps below illustrate one example.
Supplier nomination and information gathering
The process begins when a procurement team member or category manager nominates a new supplier and requests their qualification. The supplier is invited to provide information including company profile, certifications, quality management documentation, financial statements, and compliance attestations. An AI Agent can assist by validating the completeness of the supplier’s submission and flagging missing documents before the evaluation begins.
Quality and capability assessment
The supplier quality team evaluates the supplier’s manufacturing or service capabilities, quality management system, certifications (e.g., ISO 9001, GMP, AS9100), and capacity to meet requirements. If the supplier’s capabilities do not meet minimum standards, the application is returned with specific feedback or declined.
Compliance and regulatory review
Compliance reviews the supplier’s adherence to applicable regulations, industry standards, and organizational requirements such as conflict minerals, environmental compliance, labor practices, or data handling policies. An AI Agent may cross-reference the supplier’s certifications against required standards for the product or service category.
Financial stability review
Finance assesses the supplier’s financial health based on submitted financial statements, credit reports, or third-party risk data. Suppliers with financial concerns may be approved conditionally or declined. This review may proceed in parallel with the quality and compliance evaluations.
Approval decision
The evaluation results are consolidated and routed to the appropriate approver — a procurement director, supplier review board, or category manager depending on the supplier’s risk and value profile. The approver reviews the complete assessment and authorizes, conditionally approves, or declines the supplier. Conditional approvals may include corrective action requirements or limited scope authorization.
Supplier activation and record preservation
Upon approval, the supplier is added to the approved supplier list and activated in procurement systems. The complete qualification record — including all submitted documentation, evaluation results, and approval decisions — is preserved for audit and periodic re-qualification.
This process commonly relies on inputs such as the supplier application, certifications, quality documentation, financial statements, and compliance attestations. It may be triggered by a sourcing need, a new project requirement, or a category expansion. Connected systems often include procurement platforms like SAP Ariba or Coupa for supplier management, ERP systems like SAP or NetSuite for vendor master data, and third-party risk assessment tools like Dun & Bradstreet or EcoVadis.
Key decision points include whether the supplier’s quality capabilities and certifications meet minimum requirements for the intended category, whether compliance and regulatory evaluations reveal disqualifying gaps, whether financial stability is sufficient to support a reliable supply relationship, and whether the overall risk profile warrants full approval, conditional approval, or rejection.
Incomplete supplier submissions that delay the evaluation because required certifications, financial data, or compliance documentation are missing. Inconsistent evaluation criteria across categories or regions, leading to uneven supplier quality in the approved base. Prolonged sequential reviews when quality, compliance, and finance assessments happen one at a time rather than concurrently. Conditional approvals not tracked, allowing suppliers to operate without completing required corrective actions. Approved supplier records not maintained, making periodic re-qualification and audit preparation difficult.
Orchestrates supplier evaluation across procurement, quality, compliance, and finance in a single coordinated flow that engages the supplier directly.
Routes evaluations based on supplier category and risk profile so critical suppliers receive thorough assessment while lower-risk suppliers follow a streamlined path.
AI Agents validate supplier submissions at intake, flagging missing certifications, incomplete financial data, or compliance gaps before evaluation begins.
Supports parallel quality, compliance, and financial reviews so all assessment tracks proceed concurrently, reducing total qualification time.
Engages suppliers directly within the workflow so they can submit documentation, respond to questions, and receive updates in context rather than through disconnected email exchanges.
Preserves the complete supplier qualification record including all submitted documentation, evaluation results, conditions, and approval decisions for audit, re-qualification, and governance.